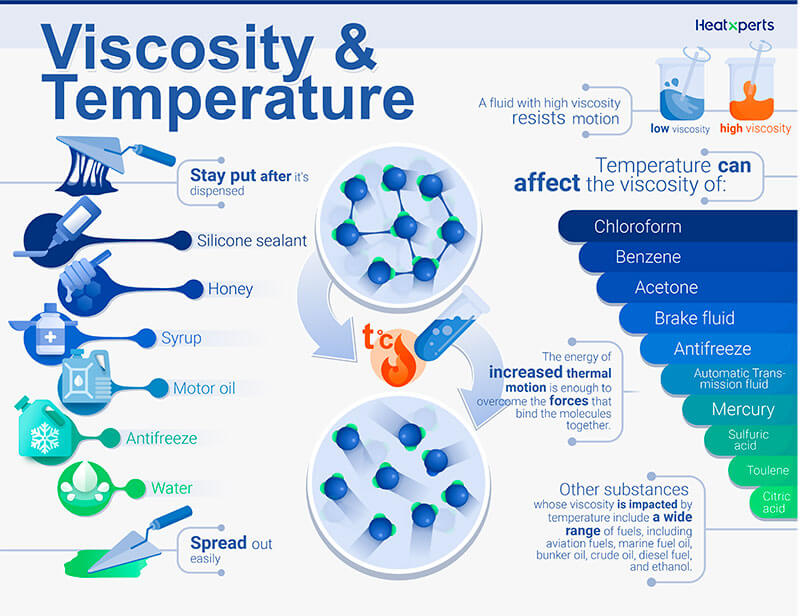 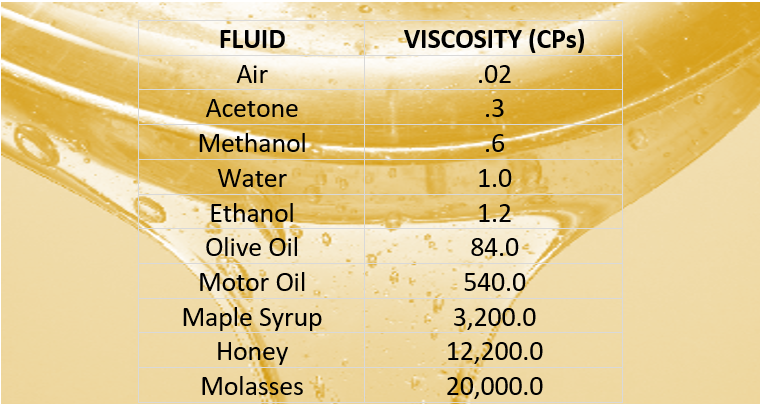
Highlighting the impact of viscosity on various applications can help readers understand its significance. "The viscosity of the oil was found to be 50 centipoise, indicating its high resistance to flow." "The viscosity of the liquid was measured in poise, a unit commonly used in fluid dynamics." This provides clarity and precision to your sentence. When discussing viscosity, it is essential to mention the units of measurement used. "Engineers consider the viscosity of a lubricant when designing machinery to ensure optimal performance." "The high viscosity of molasses makes it difficult to pour from the jar." By incorporating real-life scenarios, you can make the sentence more engaging and understandable. Using practical examples can help readers relate to the concept of viscosity. "Compared to motor oil, gasoline has a lower viscosity, allowing it to flow more easily." "Honey has a higher viscosity than water, making it flow more slowly." This allows you to emphasize the varying levels of resistance to flow. To highlight the differences in viscosity between different fluids, you can use comparative statements. "The viscosity of a fluid is influenced by the internal friction between its molecules, which determines its resistance to flow." This can help readers grasp the concept better. If you are discussing viscosity in a scientific or technical context, it is crucial to provide a more detailed explanation. "The viscosity of a fluid determines its ability to flow freely." "Viscosity is a measure of a fluid's resistance to flow." When using the word "viscosity" or the phrase "viscosity of a fluid" in a sentence, it is essential to provide a clear definition or context to ensure that the reader understands the meaning. In this article, we will explore various tips on how to use the word "viscosity" or the phrase "viscosity of a fluid" in a sentence effectively. It is a property that determines how easily a fluid can be poured or how it resists deformation when subjected to an external force. The video below shows several different long chained oils, each progressively more viscous.Viscosity is a term commonly used in the field of physics and fluid dynamics to describe the resistance of a fluid to flow. Glycerol, CH 2OHCHOHCH 2OH, is viscous partly because of the length of the chain but also because of the extensive possibilities for hydrogen bonding between the molecules. Fuel oil, lubricating grease, and other long-chain alkane molecules are quite viscous for this reason. 
This is because the molecular chains get tangled up in each other like spaghetti-in order for the liquid to flow, the molecules must first unravel. Liquids containing long molecules are invariably very viscous. 
Honey, mostly glucose and fructose (see image below) is a good example of a liquid which owes its viscosity to hydrogen bonding. Liquids whose molecules are polar or can form hydrogen bonds are usually more viscous than similar nonpolar substances. Viscosity is governed by the strength of intermolecular forces and especially by the shapes of the molecules of a liquid. Those like ether or gasoline which flow very readily have low viscosities. Liquids which flow very slowly, like glycerin or honey, have high viscosities. The resistance to such flow is called the viscosity. \)īecause its molecules can slide around each other, a liquid has the ability to flow.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
